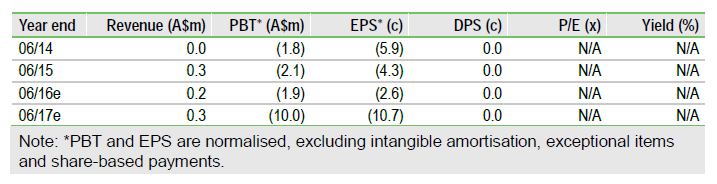
Prescient Therapeutics Ltd (AX:PTX) has sharpened its focus on lead anti-cancer compound PTX-200, and plans to add a Phase 1b trial in acute myeloid leukaemia (AML) to the ongoing Phase I/II trials in breast and ovarian cancers. It has raised ~A$2m through a share purchase plan and placement, which will fund operations until 2017.
We raise our valuation to A$49m (A$0.53/share), with the addition of the AML indication for PTX-200 more than offsetting the deferral of Phase 1b trials for PTX-100.
Developing PTX-200 a priority
Following management changes, Prescient has prioritized developing PTX-200, which is being tested in partly grant-funded, US-based Phase 1b trials in breast and ovarian cancer, including a breast cancer expansion cohort at the RPTD which began in Q116. Trials of PTX-100 have been deferred for the time being – a Phase 1b trial in breast cancer could potentially start in 2017, subject to funding.
To read the entire report, please click on the PDF file below: